Barts Health and Queen Mary launch Covid-19 research programme
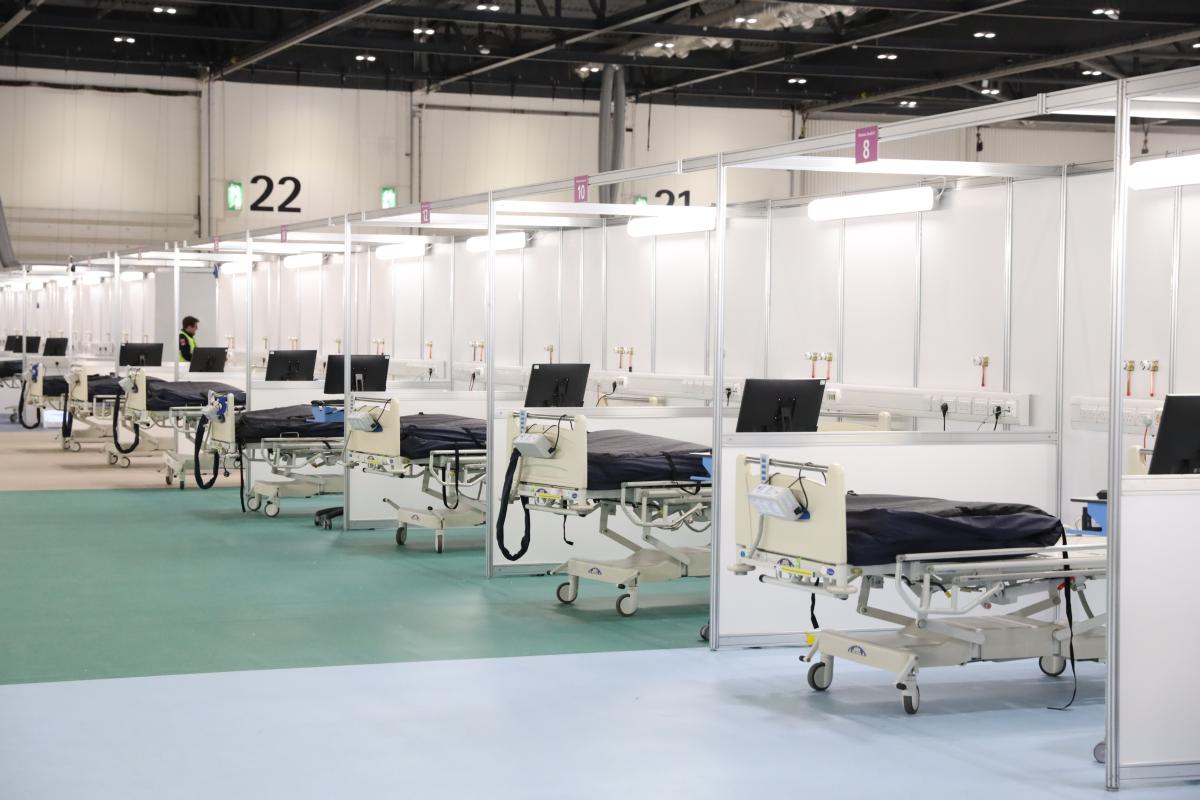
Barts Health NHS Trust and Queen Mary University of London, as part of their Barts Life Sciences initiative, have begun a new programme of Covid-19 research across Barts Health hospitals, including NHS Nightingale Hospital London.
Covid-19 patients will be recruited into clinical trials to understand why some people become severely affected by the disease, and to investigate interventions to see if they can help more patients survive.
Faced with the return to the frontline of clinical researchers to the NHS, a new team of over a hundred volunteers has been built comprising clinical researchers or scientists from other medical areas. These volunteers have been organised into teams and trained to support enrolment and delivery of COVID-19 studies for the benefit of patients.
A key consideration in studying COVID-19 patients has been how to deliver clinical trials while minimising the risk to both patients and staff. Taking studies approved by the National Institute for Health Research, the teams have developed innovative electronic consenting, processes to contact families or carers where a person is ventilated, enabling research in those most severely affected by the disease.
Just 10 days after opening the new NHS Nightingale Hospital at Barts Health, patients have already been enrolled in trials. This process, which would usually take months, has only been made possible thanks to the teamwork and courage of volunteers from across the NHS and university partners (Queen Mary, UCL and the London School of Hygiene & Tropical Medicine).
Part of the work will also look to curate a large, de-identified research data set that will create a rich data source for future covid-19 research by Queen Mary, Barts and others worldwide. Research data will be collected from patients on a daily basis to create a master data set and follow patients over time.
Professor Sir Mark Caulfield, from Queen Mary and Barts Health has been leading on COVID-19 research delivery across Barts Health hospitals, including the NHS Nightingale Hospital London.
He said: “The opening of research studies at the new Nightingale hospital in just 10 days ensures patients at all Barts Health Hospitals can get equal opportunities to participate in research. It was only possible through the amazing group of 100 plus clinical research volunteers who, at their risk, have stood shoulder to shoulder with frontline NHS staff in the fight against COVID 19 and that is what Barts Life Sciences is about.”
Dr David Collier who led clinical research training from Queen Mary and Barts Health said: “The response of over 100 volunteers, some helping from isolation at home, has enabled us to support clinical trials that would not be happening otherwise and underscores the commitment of Barts Health and Queen Mary to the community of London. As a result of their commitment and leadership, a hospital created in a conference centre has now enrolled its first research patients just days after it opened its doors.”
Barts Health and Queen Mary teams, led by Prof Rupert Pearse and Dr Mays Jawad, have expedited approval of studies with a national priority. Research teams are also being led by Professor James Moon and Professor Tom Powles.
Professor Charles Knight, Chief Executive of the NHS Nightingale Hospital London and Professor of Cardiology at Queen Mary said: “I'm deeply honoured to have been given this responsibility at a time of great pressure on the NHS and so proud that staff across Barts Health are rising to this challenge so magnificently.”
For members of the public interested in learning more about COVID-19 research taking place at Barts Health, please go to the information page.
Research staff should visit the Joint Research Management Office website, for advice and updates on conducting research during the COVID-19 outbreak.
